This resin requires the use of a Formlabs Form 3 V1 or V2
This resin requires the use of a Formlabs Form 2 LT
This resin requires post curing
This resin requires the use of a Formlabs Form 2 LT
This resin requires post curing
Biocompatible Photopolymer Resin for the Form 2 / Form 3B
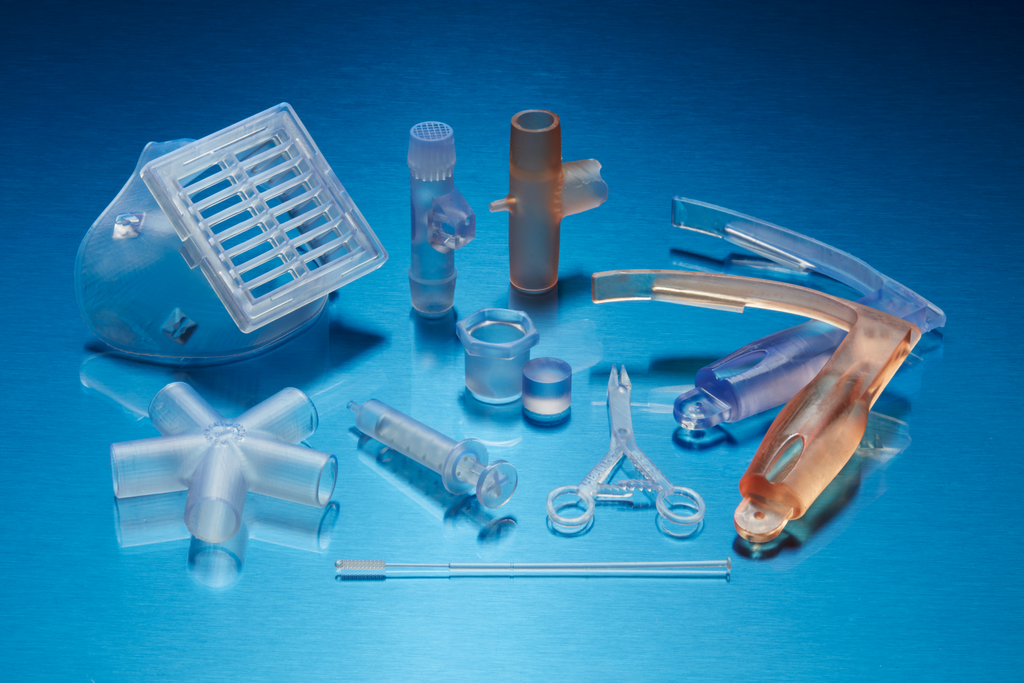
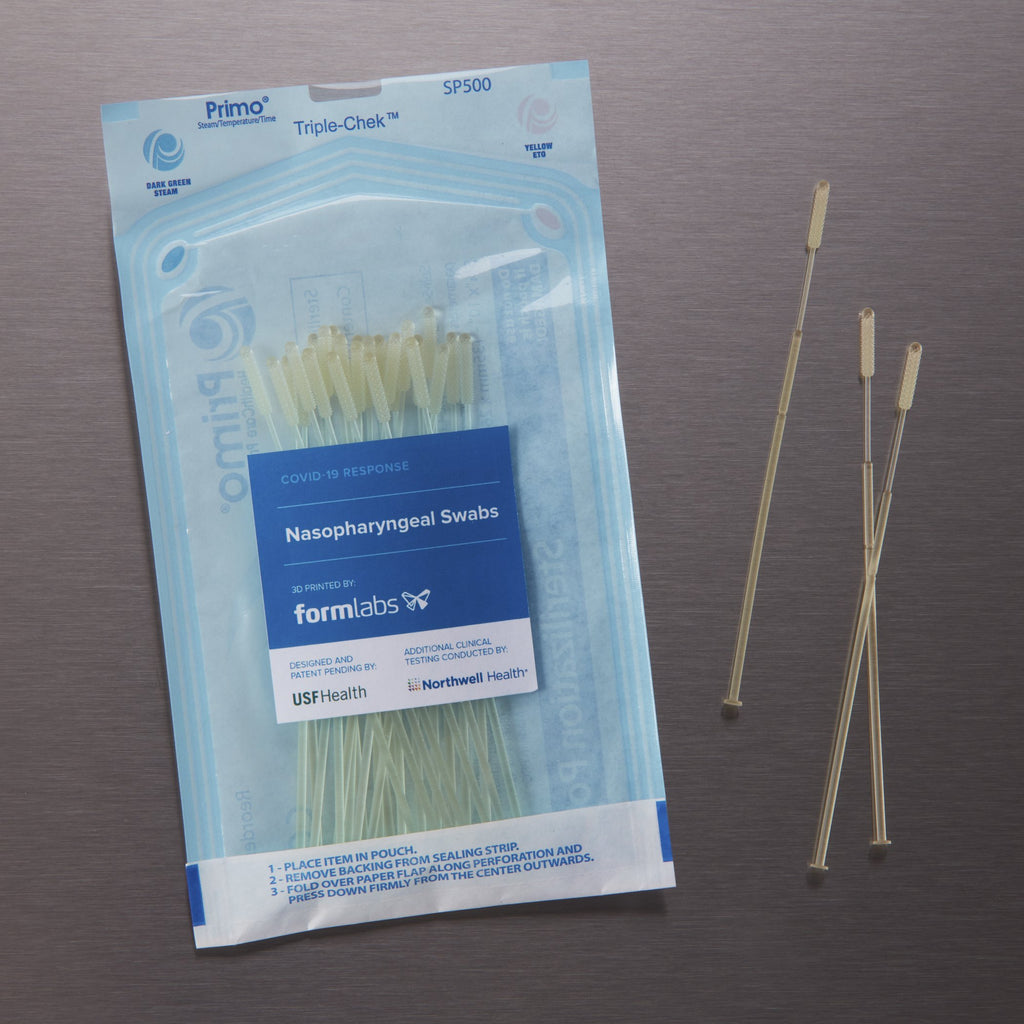
BioMed Clear Resin is a rigid material for biocompatible applications requiring long-term skin or mucosal membrane contact. This USP Class VI certified material is suitable for applications that require wear resistance and low water absorption over time.
Parts printed with BioMed Clear Resin are compatible with common sterilization methods. BioMed Clear Resin is manufactured in our ISO 13485 facility and is supported with an FDA Device Master File.

Instructions for Use | Technical Data Sheet
- Ultimate Tensile Strength: 73 MPa - 11 ksi
- Young’s Modulus: 2.9 GPa - 420 ksi
- Elongation: 12.3%
- Flexural Strength: 103 MPa - 15 ksi
- Flexural Modulus 2.5 GPa - 363 ksi
- Hardness Properties: Hardness Shore 67D
- Supports Print Resolutions: 100 and 50 microns (Form 2), 100 microns (Form 3B)
- 1 Litre of Formlabs BioMed Clear (OEM)
- Wash for 15 minutes in Form Wash
- Soak in fresh IPA for 5 minutes
- Air dry at room temperature for at least 30 minutes
- Inspect part to ensure it is clean and dry with no residual resin, IPA, or particles on the surfaces
- Cure for 60 minutes at 60 Celsius
- Chemical Disinfection - 70% Isopropyl Alcohol for 5 minutes
- Steam Sterilization: Autoclave at 134 °C for 20 minutes or Autoclave at 121 °C for 30 minutes

BioMed Clear Resin from Formlabs is a rigid and USP Class VI certified material for biocompatible applications requiring long-term skin or mucosal membrane contact. Parts printed in this material are compatible with common sterilization methods, and is suitable for manufacturing durable parts and parts requiring low water absorption.
BioMed Resins from Formlabs are biocompatible, ISO 13485 compliant, medical-grade materials developed for healthcare professionals globally. These medical materials can be used for applications that require patient skin and mucosal membrane contact.
Tested by Formlabs — All samples were printed on Form 3B SLA printers equipped with clean Build Platforms, Form 3 Resin Tanks and Biomed Amber Resin v1 cartridges. Part orientations and placement were kept constant for all samples.
All sterilization, biocompatibility, autoclave (steam), and mechanical results can be found in the Biomed Clear Sterilization Results.
BioMed Clear Resin was tested at NAMSA World Headquarters, OH, USA.

Request a Free Custom Quote
Use our easy tool to build a customized quote.
Request a Custom 3D Print Sample
Let us prove that the technology will work for your use case.
Speak with an Expert
Talk 1:1 with an expert. Tell us your challenges and our team will help you find the best solutions to meet your unique needs.